Moving forward
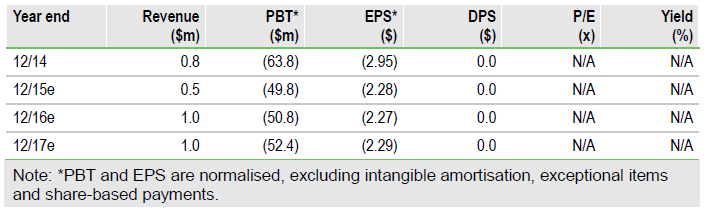
After receiving feedback from US and EU regulatory authorities, Onconova Th (NASDAQ:ONTX) submitted a trial protocol in April with overall survival as the primary endpoint for its pivotal trial in myelodysplastic syndrome (MDS) patients. We remain confident of the ultimate success of rigosertib (IV), although we have taken a more conservative view of the launch timelines. As such, we have lowered our valuation to $171m or $7.91 per basic share, from $254m or $11.70 per share.
Both regulatory bodies in April focused on high-risk MDS patients who have failed prior therapy with hypomethylating agents (HMA). The trial is likely to be enriched with the subgroups in the ONTIME trial where rigosertib (IV) demonstrated efficacy. We expect the trial to commence enrolment in H215.
Rigosertib (oral): A tale of two programmes
With Baxter deciding not to fund future trials of rigosertib (oral) as a single agent for lower-risk MDS, the future of this programme is unclear. Onconova is currently awaiting data to determine which patients are most likely to respond before announcing its next steps. These data may be available by year end. However, combination data with azacitidine in MDS/AML patients continue to be positive.
To Read the Entire Report Please Click on the pdf File Below