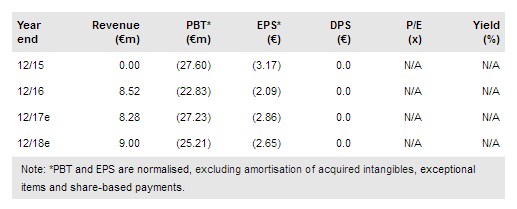
The development of Celyad’s (NASDAQ:CYAD) natural killer (NK) receptor-based CAR T-cell therapy (NKR-2/CYAD-01) has taken an important step with the initiation of the SHRINK trial, which moves NK CAR T-cell therapy towards the 90%+ of cancer patients who do not have CD19 or BCMA tumours. CYAD-01 is already being tested in THINK with five solid tumour types plus AML and MM. Promising THINK results have been reported at the lowest dose. Celyad has paid $25m in cash and shares to reduce the royalties payable on potential short-term deals and long-term sales. Our indicative value of Celyad has been revised to €524m or €51.6 per share.
THINK: Two stable disease cases and revised deal
Celyad is running the large Belgian and US immuno-oncology autologous CYAD-01 THINK trial. Celyad has reported two cases of stable disease after three months at the first 3x108 dose level in previously progressing colorectal patients. The second 1x109 dose cohort is currently being treated. Overall data (six-month) are possible in H218. Tumours showing efficacy could then move into expanded studies allowing BLA filings from 2020.
To read the entire report Please click on the pdf File Below: