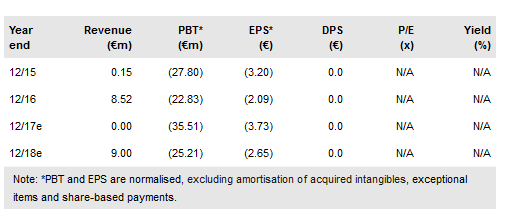
Celyad SA (NASDAQ:CYAD) has provided an update on its trial plans and announced 2016 preliminary results. The THINK Phase Ib trial is a major expansion of CAR therapy with five solid tumours plus AML and MM being explored. The THINK dose escalation results are expected in Q417 with six-month efficacy results possible from H218. The colorectal, SHRINK trial starting in Q2 will explore combining NKR-2 therapy with chemotherapy. The Q3 LINK trial will explore direct delivery of NKR-2 cells to metastatic liver tumours. The move into solid tumours puts Celyad in a leading position. Our interim indicative value remains at €45 per share. Cash remains strong at €82.6m.
NKR-2 moving through dose escalation
Celyad is now running both Belgian and US arms of the immuno-oncology autologous NKR-2 trials (THINK). The first dose cohorts have been recruited. Data on the final dose (up to 3bn (3 x 109) cells is expected in Q417. One arm has two haematological cancers: AML and MM. The other has five solid tumours: colorectal, triple negative breast, pancreatic, bladder and ovarian. At the highest dose, each tumour type then moves into a 14-patient efficacy phase. Data (six-month) are possible from H218. Tumour types showing efficacy could then move directly into expanded studies allowing BLA filings, perhaps from 2019-20. Other CAR companies are in the congested CD19 area with few trials in solid tumours.
To read the entire report Please click on the pdf File Below