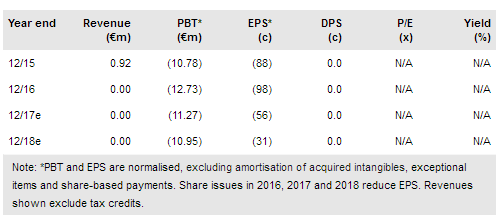
Txcell (PA:TXCL) has agreed an important manufacturing supply contract with Lentigen, a German-US producer of the critical lentivirus reagent essential for modifying T-cells. This is a core element for the development of CAR-modified regulatory T-cells (CAR Treg). Production of any modified T-cell therapy is complex and companies need to be able to scale up production and keep costs and prices under control. In 2018, warrants could bring a further €10.8m in cash covering costs until the IND is filed for the first ever CAR Treg clinical trial. Our indicative valuation remains at €74m.
Focus on CAR Treg with transplant in the lead
TxCell is now focused on the CAR Treg ENTrIA platform technology (see note of 28 February 2017) using chimeric antigen receptor (CAR) technology similar to that in the CAR T-cell cancer area; TxCell holds a core granted US patent. ENTrIA needs viral vectors to deliver genes to isolated T-cells. This process needs to be finalised before the first clinical CAR Treg trial – due in late 2018 for transplant. Other potential indications are lupus nephritis and bullous pemphigoid (skin); TxCell has a research collaboration on multiple sclerosis with INSERM and Nantes University.
To read the entire report Please click on the pdf File Below: