RAPPER II trial off to a good start
RAPPER II is a 100-patient study to measure the time taken for wheelchair users to mobilise in a Rex Bionics PLC (LONDON:RXB) unit and complete an exercise regime. The study also intends to provide a better understanding of the severity of injury that can be safely treated with a Rex unit. The first patient has successfully completed the assessment and the data from the first cohort of patients are expected to be presented before the end of 2015. Trials such as this and RAPPER I, are key to driving adoption and therefore although it is still in its early stages, it is encouraging to see the trial get off to a good start.
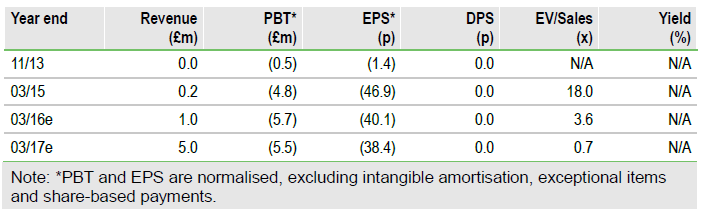
2.1m. A further £1.0m may be raised from a number of specific investors before a general meeting to approve the placing. There was £4.4m cash on the balance sheet at 31 March 2015 and approximately £420k per month is being spent, so these placings would bring the estimated cash balance at the end of June to £6.2m assuming £2.9m is raised net of costs. A general meeting will be held on 30 June 2015 to approve the transaction. Annual results were reported at the end of May and were in line with our forecasts, although at this stage the progress of the trials and commercial agreements is more important than periodic earnings.
Global distribution agreements being put in place
Rex has already made good progress in finding distributors, and has signed agreements with Deltason in Hong Kong, Blue-Ocean Robotics in Denmark and Ortho-Medico in Benelux. It also has a short list of potential partners for US and China which are two important potential markets. Successful outcomes from trials are still key to driving uptake, but having a distribution network in place is important to maximise the sales potential as demand develops.
To Read the Entire Report Please Click on the pdf File Below