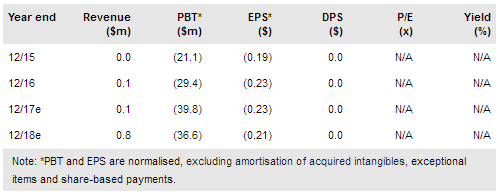
Redhill Biopharma Ltd (NASDAQ:RDHL) delivered a flurry of news recently, including a major R&D milestone when a Phase III trial with BEKINDA 24mg showed that the drug was beneficial to gastroenteritis/gastritis patients. There are several inflection points still to come in 2017: a data readout from the BEKINDA Phase II trial with IBS-D patients, a second DSMB review of RHB-104 Phase III in Crohn’s disease trial with an early termination option, and the meeting with the FDA to decide the further strategy with BEKINDA for gastroenteritis. We have increased our valuation to $390m (NIS1.42bn).
R&D milestone: Successful Phase III with BEKINDA
On 14 June 2017, RedHill announced positive top-line results from its GUARD Phase III trial with gastroenteritis patients receiving BEKINDA 24mg, which is a once-daily, bi-modal release, oral formulation of antiemetic drug ondansetron. The primary endpoint was met with statistical significance and, as RedHill indicated, it will know whether another Phase III trial is needed after its meeting with the FDA, expected by October 2017. If approved, it could be the first once-daily formulation of ondansetron for acute gastroenteritis to reach the US and European markets.
To read the entire report Please click on the pdf File Below: