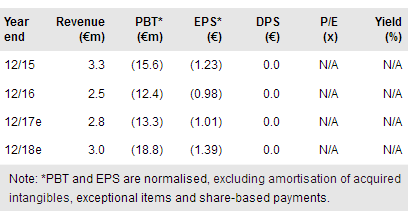
Pixium Vision (PA:PIX) has received clearance from the French regulatory agency (Agence Nationale de Sécurité du Médicament et des Produits de Santé, or ANSM) to start a feasibility study of its Prima sub-retinal bionic vision system (BVS) on up to five patients with advanced Dry-ARMD. It plans to complete the first implantation before YE17, and obtain six-month study data in H218. If results are positive, Pixium expects to start an EU pivotal study in H119. We have reinstated our Prima probability of success estimate in our model to 12.5% (from 10%), leading to an increase in our pipeline rNPV to €82.6m (from €63.0m, previously).
Feasibility trial on patients with severe Dry-ARMD
The study will take place at Fondation Ophtalmologique Rothschild and Hôpital des Quinze-Vingt in Paris, and recruit up to five patients with advanced dry age-related macular degeneration (Dry-ARMD). Eligible patients will be required to have the most severe form of the disease, including a visual acuity (VA) of 20/400 or worse, with no light perception in the foveal (central portion of the retina) region, and an area of central retinal atrophy of at least 3-disc-diameters (approximately 5mm in length). If six-month results (expected H218) are positive, Pixium plans to start an EU pivotal study in early 2019. Pixium is currently also in discussions with the FDA and aims to obtain approval by YE17 to start a US feasibility study.
To read the entire report Please click on the pdf File Below: