Myriad Genetics, Inc. (NASDAQ:MYGN) recently announced the expansion of companion diagnostic (CDx) partnership with AstraZeneca (NYSE:AZN) and Merck & Co., Inc. (NYSE:MRK) .
The companies have again come together to use BRACAnalysis CDx to find germline BRCA mutations in men with metastatic castrate-resistant prostate (mCRPC) cancer enrolled in the Phase III PROfound (NCT02987543) study.
On successful completion of the study, Myriad Genetics plans to file a supplementary premarket approval application with the FDA for using BRACAnalysis CDx as a CDx to Lynparza (olaparib) in the patient population mentioned above.
Market Potential
Per the American Cancer Society, around 174,650 new cases of prostate cancer are estimated to be diagnosed in the United States in 2019. Moreover, 42,970 new cases of mCRPC are expected to get registered in 2019. As a matter of fact, prostate cancer is considered the second principal cause of cancer death in American men. Hence, we believe that the recent development is a timely one and will help Myriad Genetics boost top-line contributions in the near term.
Regulatory Tidings for BRACAnalysis
The partnership between Myriad Genetics and AstraZeneca on Lynparza had commenced in 2007. Since then, BRACAnalysis CDx has received multiple regulatory approvals. In February 2019, the Japanese Ministry of Health, Labour, and Welfare approved the use of BRACAnalysis CDx as a companion diagnostic to discover patients with germline BRCA mutated (BRCAm) advanced ovarian cancer and eligible for first-line maintenance therapy with Lynparza.
Last December, the company announced FDA approval for the use of BRACAnalysis CDx test to identify patients with germline BRCA mutation, suffering from advanced ovarian cancer and suitable for first-line maintenance therapy with AstraZeneca’s PARP (poly ADP (NASDAQ:ADP) ribose polymerase) inhibitor — Lynparza — following response to platinum-based chemotherapy.
Last March, the Japanese Ministry of Health, Labour, and Welfare approved BRACAnalysis CDx as a companion diagnostic to identify patients with germline BRCAm metastatic breast cancer, treated previously with chemotherapy and eligible for treatment with Lynparza.
In January 2018, the FDA approved BRACAnalysis CDx as a companion diagnostic for the same as above.
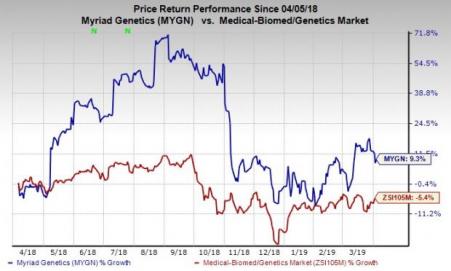
Price Performance
Over the past year, Myriad Genetics’ share price has outperformed the industry. The stock has gained 9.3% against the industry’s 5.4% decline.
Zacks Rank & Stocks to Consider
Myriad Genetics currently carries a Zacks Rank #3 (Hold).
A few better-ranked stocks in the broader medical space are Penumbra, Inc. (NYSE:PEN) .
Penumbra’s long-term earnings growth rate is expected at 20.9%. The stock presently carries a Zacks Rank #2 (Buy). You can see the complete list of today’s Zacks #1 Rank (Strong Buy) stocks here.
Zacks' Top 10 Stocks for 2019
In addition to the stocks discussed above, would you like to know about our 10 finest buy-and-holds for the year?
Who wouldn't? Our annual Top 10s have beaten the market with amazing regularity. In 2018, while the market dropped -5.2%, the portfolio scored well into double-digits overall with individual stocks rising as high as +61.5%. And from 2012-2017, while the market boomed +126.3, Zacks' Top 10s reached an even more sensational +181.9%.
See Latest Stocks Today >>
Merck & Co., Inc. (MRK): Free Stock Analysis Report
AstraZeneca PLC (AZN): Free Stock Analysis Report
Penumbra, Inc. (PEN): Free Stock Analysis Report
Myriad Genetics, Inc. (MYGN): Free Stock Analysis Report
Original post
Zacks Investment Research
