Eli Lilly and Company (NYSE:LLY) and Incyte Corporation (NASDAQ:INCY) announced positive top-line data from two late-stage studies evaluating their oral JAK inhibitor, Olumiant (baricitinib), as monotherapy for moderate-to-severe atopic dermatitis (“AD”), a type of eczema. Data from the phase III studies – BREEZE-AD1 and BREEZE-AD2 – showed that the candidate met the primary endpoints of statistically significant improvements in patients compared to placebo as measured by Investigator's Global Assessment for AD (“IGA”) score after treatment of 16 weeks.
Detailed data from these studies along with ongoing phase III studies on barcitinib will be presented and published at future scientific venues and in peer-reviewed journals later in 2019.
Olumiant was first approved in early 2017 for treating moderately-to-severely active rheumatoid arthritis (“RA”) in patients with inadequate response or intolerant to TNF inhibitor therapies in Europe. In June 2018, Olumiant received approval in the United States for similar indication. However, the FDA allowed only the lower dose of the drug.
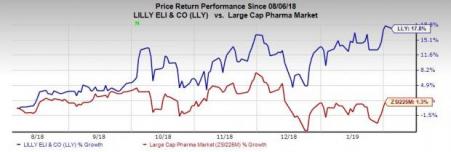
In the past six months, Lilly’s shares have outperformed the industry. The stock gained 17.8% while the industry rose 1.3% in the said time frame.
Olumiant is one of the drugs in Lilly’s new pharmaceutical product portfolio, which is anticipated to be a key top-line driver going ahead. The drug generated $132.5 million in the first nine months of 2018. Although sales in Europe were upbeat for the drug on the back of launch in new markets, U.S. sales were weak in the third quarter. Olumiant’s fourth-quarter sales may provide an idea about the drug’s demand in the country.
Lilly is also developing Olumiant as treatment for systemic lupus erythematosus and alopecia areata in mid-to-late stage studies.
Meanwhile, competition is on the rise in the JAK inhibitor segment as well as the eczema market with several approved as well as under-development therapies from several companies. Regeneron Pharmaceuticals and Sanofi’s Dupixent is approved for treating AD since 2017 while AbbVie (NYSE:ABBV) and Pfizer (NYSE:PFE) are developing their respective candidates for the indication in late-stage studies..
Zacks Rank
Lilly currently carries a Zacks Rank #2 (Buy). You can see the complete list of today’s Zacks #1 Rank (Strong Buy) stocks here.
Zacks' Top 10 Stocks for 2019
In addition to the stocks discussed above, would you like to know about our 10 finest buy-and-holds for the year?
Who wouldn't? Our annual Top 10s have beaten the market with amazing regularity. In 2018, while the market dropped -5.2%, the portfolio scored well into double-digits overall with individual stocks rising as high as +61.5%. And from 2012-2017, while the market boomed +126.3, Zacks' Top 10s reached an even more sensational +181.9%.
Eli Lilly and Company (LLY): Free Stock Analysis Report
Pfizer Inc. (PFE): Get Free Report
AbbVie Inc. (ABBV): Free Stock Analysis Report
Incyte Corporation (INCY): Get Free Report
Original post