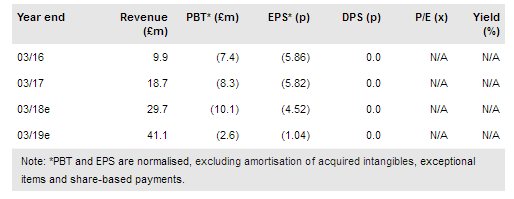
Abzena PLC (LON:ABZA) has announced another licensing deal for its proprietary site-specific ThioBridge antibody drug conjugate (ADC) linker technology. This time it is with a Taiwanese biopharmaceutical company (OBI Pharma). The agreement enables the development of OBI Pharma’s proprietary ADC, OBI-999. The deal also allows OBI to develop further ADCs as potential treatments for cancer and includes a master services and clinical supply agreement. This again provides important validation of Abzena’s ADC technology and hybrid business model. Our rNPV valuation increases slightly to £134m (vs £132m) following inclusion of one ADC being developed through this deal and we note there is potential for more.
Another ThioBridge licensing deal
Abzena has announced a licensing deal with OBI Pharma, a Taiwanese biopharmaceutical company for its novel site-specific ThioBridge technology, which links antibodies/proteins to cytotoxic agents. Alongside this, both parties have also entered into a master services and clinical supply agreement for Abzena to provide further manufacturing process development and GMP manufacture of OBI-999 and other ADCs as they are developed. According to the company, the value of the agreement has the potential to reach up to £128m in development/commercial milestones if the partner successfully develops the ADC products. In addition, Abzena would also receive royalties on sales of any approved products that incorporate the ThioBridge technology.
To read the entire report Please click on the pdf File Below: